Biomolecules are the complex organic molecules which build up living organism and are essential for their growth and maintenance. For examples proteins, carbohydrates, lipids and nuclic acids.
Carbohydrates:
Carbohydrates are a class of naturally occurring organic compounds of carbon, hydrogen and oxygen. These contain the polyhydric aldehydes, polyhydric ketones and large polymeric substances which can be broken down to polyhydric aldehydes and ketones. These include glucose, fructose, cellulose, sucrose, starch, etc.
Classification of Carbohydrates: Carbohydrates may be classified into:
- Sugars: These are crystalline substances which are sweet and water soluble. e.g. glucose, fructose and cane sugar.
- Non-sugar: These are tasteless, insoluble in water and amorphous. e.g. starch, cellulose, etc.
Carbohydrates are also classified as–
- Monosaccharide: These are the simplest carbohydrates which cannot be hydrolysed into simpler compounds. Therefore, they represent the simplest single carbohydrate units. The general formula of (CH2O) n. If the carbohydrates contain aldehydic group, they are named as aldose and if they have ketonic group, they are called ketoses. The common examples are ribose (C5H10O5), glucose (C6H12O6), fructose (C6H12O6) etc.
- Oligosaccharides: These are carbohydrates which give two to ten monosaccharide molecules on hydrolysis. These are further classified into disaccharides, trisaccharides, tetrasaccharides, etc. depending upon the number of monosaccharide units present in their molecules. e.g. Disaccharides: Sucrose, lactose, maltose. All these have the molecular formula C12H22O11.Trisaccharides: raffinose (C18H32O16).Tetrasaccharides: stachyose (C24H42O21).
- Polysaccharides: These are carbohydrates which are polymeric and can be hydrolysed to give a large number of monosaccharide units. e.g. cellulose, starch, glycogen, etc. The general formula of starch and cellulose is (C6H10O5) n. These get hydrolysed to give monosaccharides.
(C6H10O5) n + nH2O ———→ n C6H12O6
Starch / cellulose Glucose
Monosaccharides:
Monosaccharides are polyhydric aldehydes and ketones which cannot be hydrolysed into simpler carbohydrates. The monosaccharides with an aldehydic group (—CHO) are called aldoses while those with ketonic group (—C = O) are called ketoses. They contain up to six carbon atoms. The general formula of (CH2O) n. They are further classified as trioses, tetroses, pentoses, hexoses, etc. depending upon the number of carbon atoms they contain. Some examples of various categories of monosaccharides are given below:
3 Aldotrioses: C3H6O3 CH2OH CHOH CHO Glyceraldehyde
4 Aldotetroses: C4H8O4 CH2OH (CHOH)2 CHO Erythrose, Threose
5 Aldopentoses: C5H10O5 CH2OH (CHOH)3 CHO Arabinose,Ribose,Xylose, Lyxose
6 Aldohexoses: C6H12O6 CH2OH (CHOH)4 CHO Glucose, Galactose, Mannose, Allose, Talose, Gulose, Idose etc.
3 Ketotrioses: C3H6O3 CH2OH CO CH2OH Dihydroxyacetone
4 Ketotetroses: C4H8O4 CH2OH CO CHOH CH2OH Erythrulose
5 Ketopentoses: C5H10O5 CH2OH CO (CHOH)2 CH2OH Ribulose, Xylulose
6 Ketohexoses: C6H12O6 CH2OH CO (CHOH)3 CH2OH Fructose, Sorbose, Tagatose, etc.
Reducing and non-reducing sugars:
Those saccharides which reduce Fehling’s reagent and Tollen’s reagent are called reducing sugars, and those which do not reduce, they are called non-reducing sugar.
Polysaccharides:
These are neutral polymeric compounds in which a large number of monosaccharide units are joined by glycosidic linkages. They have the general formula (C6H10O5) n, where n has very large value. They are colourless, tasteless and are insoluble in plant and animal life as food storage and structural role. They are usually made up of pentoses or hexoses. The important polysaccharides are cellulose, starch and glycogen.
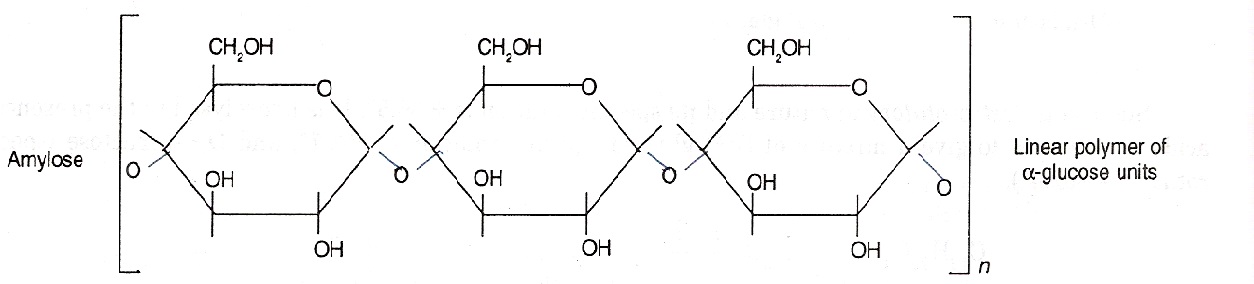
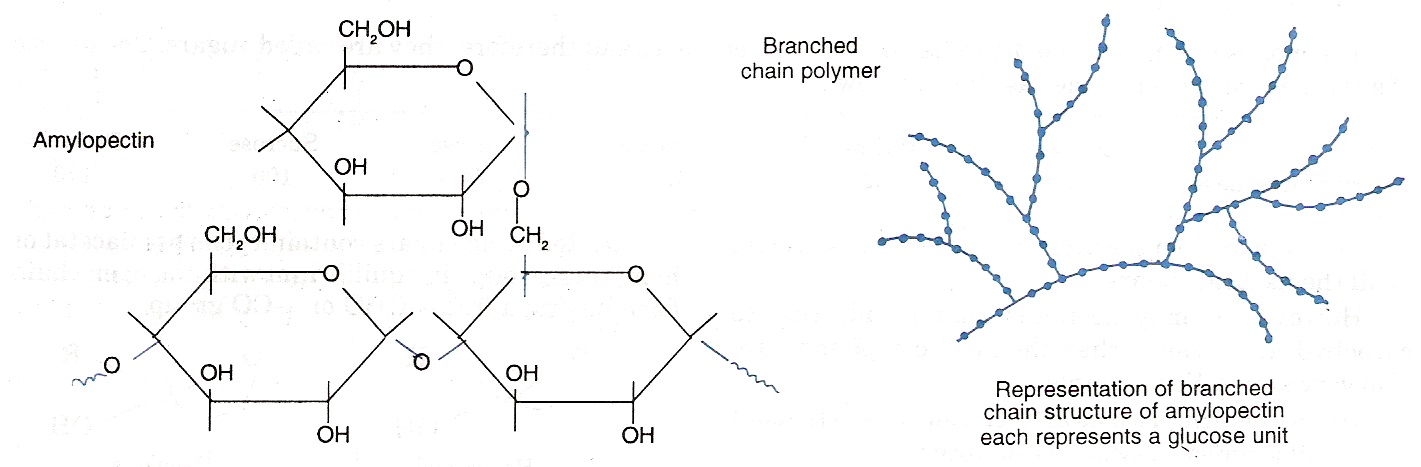
- Starch: Starch is the main storage polysaccharide of plants. It is a polymer of α-glucose and consists of two components namely amylose and amylopectin.
- Amylose is a water soluble fraction. It is a linear polymer of α-glucose as shown below:

- Amylopectin is water insoluble fraction. It is consists of branched chains of α-glucose as shown below. For simplicity, it may represented as-
 Starch is used as the principal food storage of glucose energy. It comprises the major part of roots, seeds and corns.
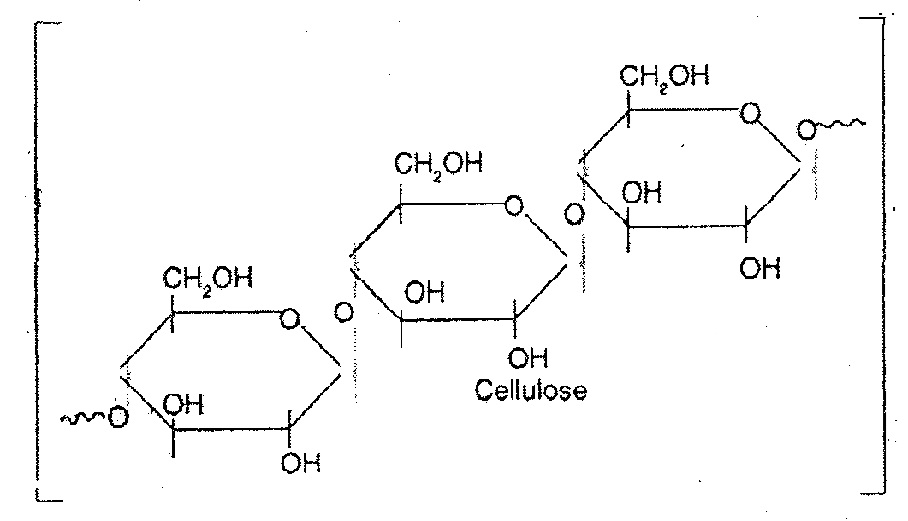
Starch is used as the principal food storage of glucose energy. It comprises the major part of roots, seeds and corns. - Cellulose: It is also major structural polysaccharide in higher plants where it constitutes the bulk of the cell wall. It is probably the most abundant organic substance found in nature. Over 50% of the total organic matter in the living world is cellulose. Structurally, its molecules are made up of a large number of β-glucose units. The chains are arranged to form bundles and are held together by hydrogen bonds between glucose of adjacent strands.

 Starch is used as the principal food storage of glucose energy. It comprises the major part of roots, seeds and corns.
Starch is used as the principal food storage of glucose energy. It comprises the major part of roots, seeds and corns.