Depression in freezing Point:
The freezing point is the temperature at which the solid and the liquid states of the substance have the same vapour pressure. It has been observed that when a non-volatile solute is added to a solvent, the freezing point of the solution is always lower than that of the pure solvent. It may be illustrated graphically as shown below:
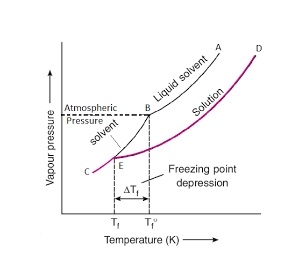
In the figure, the curve BC gives the vapour pressure of the pure solvent. We know that the addition of a non-volatile solute lowers the vapour pressure and the curve DE gives the vapour pressure curve for the solution at different temperatures. The curve AB corresponds to the vapour pressure of the solid at different temperatures. The temperature corresponding to the point B where the solid and liquid solvent meet (i.e., solid and liquid states have the same vapour pressure) represents the freezing point temperature of pure solvent (Tf°). The temperature corresponding to the point B where the solid solvent and liquid solution meet (i.e., solid and liquid states have the same vapour pressure) represents the freezing point temperature of the solution (Tf).
The difference of the freezing point of pure solvent and the solution is called the depression of freezing point, it is denoted by ΔTf . Therefore,
ΔTf = Tf° – Tf
It has been determined experimentally that the depression of freezing point of a solution is proportional to the molal concentration of the solution. Thus,
ΔTf α m
Or, ΔTf = Kf m ———> (2)
Where m is the molality of the solution and represents moles of solute in 1 kg of solvent and Kf is called molal depression constant.
If m = 1, then
ΔTf = Kf
Thus, molal depression constant, Kf, is defined as ‘the depression of freezing point produced when one gram mole of solute dissolved in 1 kg (or1000 g) of the solvent.
Again, we know
Molality (m) = (WA /Mol. mass) x (1000/WB )
Putting the value of m in expression (2) we have,
ΔTf = (Kf x WA x 1000) / (WB x MA)
Where, WA = Mass of solute, MA = Molecular mass of solute, WB = Mass of solvent, Kf = Molal depression constant & ΔTf = Depression of freezing point.