Oxidation Number:
Oxidation number is a number that represents an electric charge which an atom has or appears to have when combined with other atoms. An atom can have positive, zero or negative oxidation number.
In order to know the oxidation number, the electrons are counted according to the following two rules:
1) For homonuclear diatomic molecules, for examples, H2, Cl2, N2, O2, etc electrons shared between two similar atoms are divided equally between the sharing atoms. For example, in hydrogen molecule (H2), the electron pair is equally shared between the two hydrogen atoms. Therefore, one electron is counted with each hydrogen atom as shown below:
H׃H
The net charge on each atom of hydrogen is equal to zero, because the number of electron (one) is equal to number of proton (one). Hence, oxidation number of H2 molecule is zero.
2) For hetero-nuclear molecules for example HF, HCl, H2O etc. The electrons shared between two unlike atoms are shifted towards more electronegative atoms. For example, in case of hydrogen chloride molecule, chlorine is more electronegative than hydrogen. Therefore, the shared pair is counted towards chlorine atom as shown below:
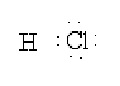 As a result, chlorine gets one extra electron and acquires a unit negative charge. Hence, oxidation number of chlorine is -1. On the other hand, hydrogen atom without electron has a unit positive charge. Hence, oxidation number of hydrogen in hydrogen chloride is +1.
As a result, chlorine gets one extra electron and acquires a unit negative charge. Hence, oxidation number of chlorine is -1. On the other hand, hydrogen atom without electron has a unit positive charge. Hence, oxidation number of hydrogen in hydrogen chloride is +1.
Keeping in view, the above concept, following rules are given for the determination of oxidation number of an atom in a molecule.
1) The oxidation number of any substance in its elementary state is always zero. For example, oxidation number of H2, Cl2, N2, O2, S, Zn, etc is always zero.
2) The oxidation number of the element in monoatomic ion is equal to the charge on the ion. For example, in K+ Cl–, the oxidation number of K is +1 while that of Cl is -1. In the similar way, oxidation number of all alkali metals is + 1 while that of alkaline earth metals are + 2 in their compounds.
3) The oxidation number of fluorine is always -1 in all its compounds. Other halogens (Cl, Br and I) also have an oxidation number of -1, when they occur as halide ions in their compounds.
4) Hydrogen is assigned oxidation number +1 in all its compounds except in metal hydrides. In metal hydrides like NaH, MgH2, CaH2, LiH, etc., the oxidation number of hydrogen is -1.
5) Oxygen is assigned oxidation number -2 in most of its compounds, however, in peroxides (which contain O—O linkage) like H2O2, BaO2, Na2O2, etc. its oxidation number is -1. Similarly, the exception also occurs in compounds of fluorine and oxygen like OF2 and O2F2 in which the oxidation number of oxygen is +2 and +1 respectively.
6) Oxidation number of neutral molecules like HCl, NH3, CH4, etc is zero.
7) For complex ions, e.g. NH4+, PO4-3, SO4-2, etc the oxidation number is equal to the charge on the ions.
Calculation of Oxidation number:
1) Find the oxidation number of Cr in the following compounds: K2CrO4, K2Cr2O7 and Cr2O7-2
(i) Let ‘X’ be the oxidation number of Chromium in K2CrO4.
We Know,
Oxidation number of K = +1
Oxidation number of O = -2.
Therefore,
K2CrO4 = 0
Or, (+1) x 2 + X + (−2) x 4 = 0
Or, 2 +X −8 = 0
Or, X − 6 = 0
X = +6
Therefore, oxidation number of Cr in K2CrO4 is +6.
(ii) Let ‘X’ be the oxidation number of Chromium in K2Cr2O7.
We Know,
Oxidation number of K = +1
Oxidation number of O = -2.
Therefore, K2Cr2O7 = 0
Or, (+1) x 2 + 2X + (−2) x 7 = 0
Or, 2 +2X −14 = 0
Or, 2X − 12 = 0
Or, X = 12/2
X = +6
Therefore, oxidation number of Cr in K2CrO4 is +6.
(iii) Let ‘X’ be the oxidation number of Chromium in Cr2O7-2.
We Know,
Oxidation number of K = +1
Oxidation number of O = -2.
Therefore, Cr2O7= −2
Or, 2X + (−2) x 7 = −2
Or, 2X −14 = −2
Or, 2X = 14 −2
Or, X = 12/2
X = +6
Therefore, oxidation number of Cr in CrO7−2 is + 6.
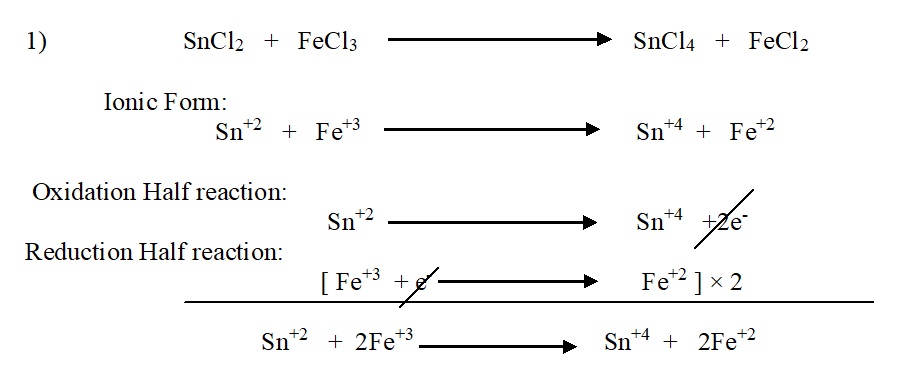
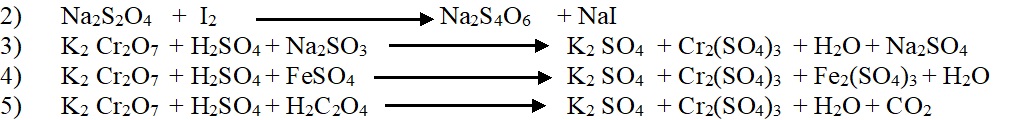
Balancing of chemical equation by electron ion methods: