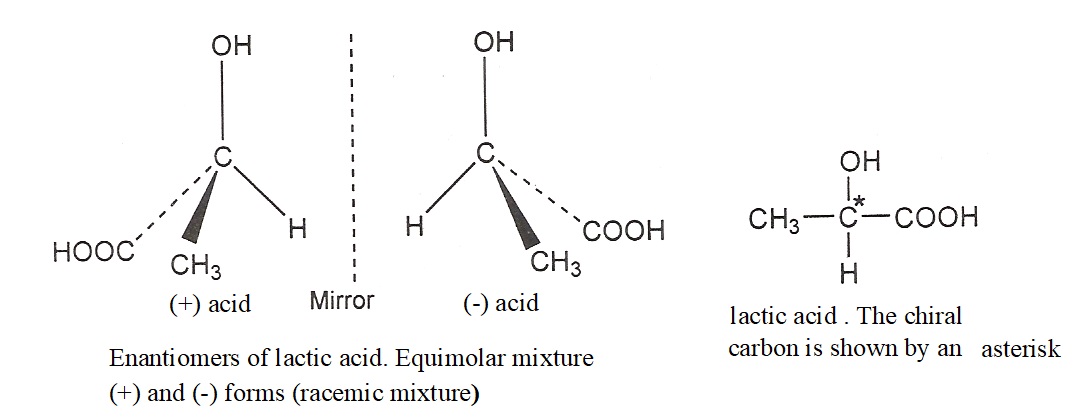
An equimolecular mixture of a pair of enantiomers is called racemic mixture. A racemic mixture is optically inactive because of the fact that in equimolecular mixture of enantiomeric pairs, the rotation caused by the molecules of one enantiomer is cancelled by the rotation of other enantiomers. Therefore, racemic mixture becomes optically inactive because of external compensation. For example, racemic mixture of lactic acid is represented by (±) lactic acid.
Lactic acid (2-Hydroxypropanoic acid) is an example of a compound which shows optical isomerism as it contains one chiral carbon atom. Two, three dimensional structures are possible for lactic acid. These structure are not identical because they cannot be superimposed on each other. One is the mirror image of other. Such nonsuperimposable mirror image forms are optical isomers and are called enantiomer. These are shown below.
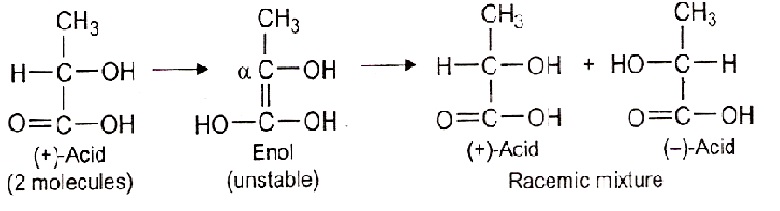
Racemisation:
Racemisation is a process of conversion of an optically active compound into the racemic mixture. Both (+) and (-) forms of the compound are capable of racemisation under the influence of heat, light or chemical reagent. e.g. increase in temperature favours racemisation of lactic acid, tartaric acid etc give racemic mixture on heating.