Shape of orbitals:
An orbital is the region of space around the nucleus within which the probability of finding electron of a given energy is maximum. The shape of this region (electron cloud) gives the shape of the orbitals. Let us consider the individual shapes-
Shape of s-orbital:
For s-orbital, when l=0, the value of m is zero i.e. there is only one possible orientation. This means that the probability of finding electron is the same in all direction at a given distance from the nucleus. It should be spherical in shape because a sphere can be defined completely by a single value i.e. its radius. Hence, all s-orbital are non-directional and spherically symmetrical about the nucleus.
The size of an s-orbital depends upon the value of principal quantum number (n); greater the value of ‘n’ larger is the size of the orbital. Therefore, 2s-orbital is larger than 1s-orbital, but both of them are non-directional and spherically symmetrical shape.
An important features of the 2s-orbital is that there is a spherical shell within this orbital where the probability of finding electron is zero. This is called Node or Nodal surface.
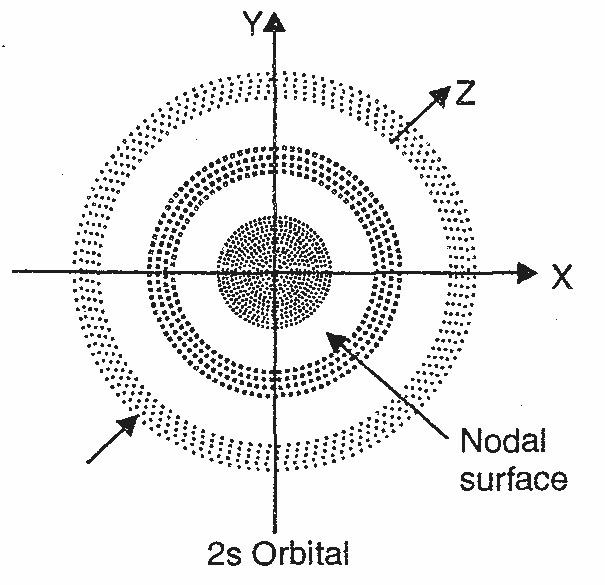
Fig: Shape of s- orbital
Shape of p-orbital:
For p-subshell, l=1, there are three values of m viz. -1, 0, +1. It means p-orbital can have three possible orientations. These are equal in energy (degenerate state) but differ in their orientations. Each p-orbital consist of two lobes symmetrical about a particular axis. Depending upon the orientation of the lobes, these are denoted by the symbol px, py and pz accordingly as they are symmetrical about x, y, and z.
The two lobes of each p-orbital are separated by a nodal plane (a plane having zero electron density). Thus, p-orbitals are dumbbell shape and have directional characters. The probability of finding electron is equal in both the lobes.
Notes: The p-orbitals of higher energy level have similar shapes although their shape, sizes are bigger.
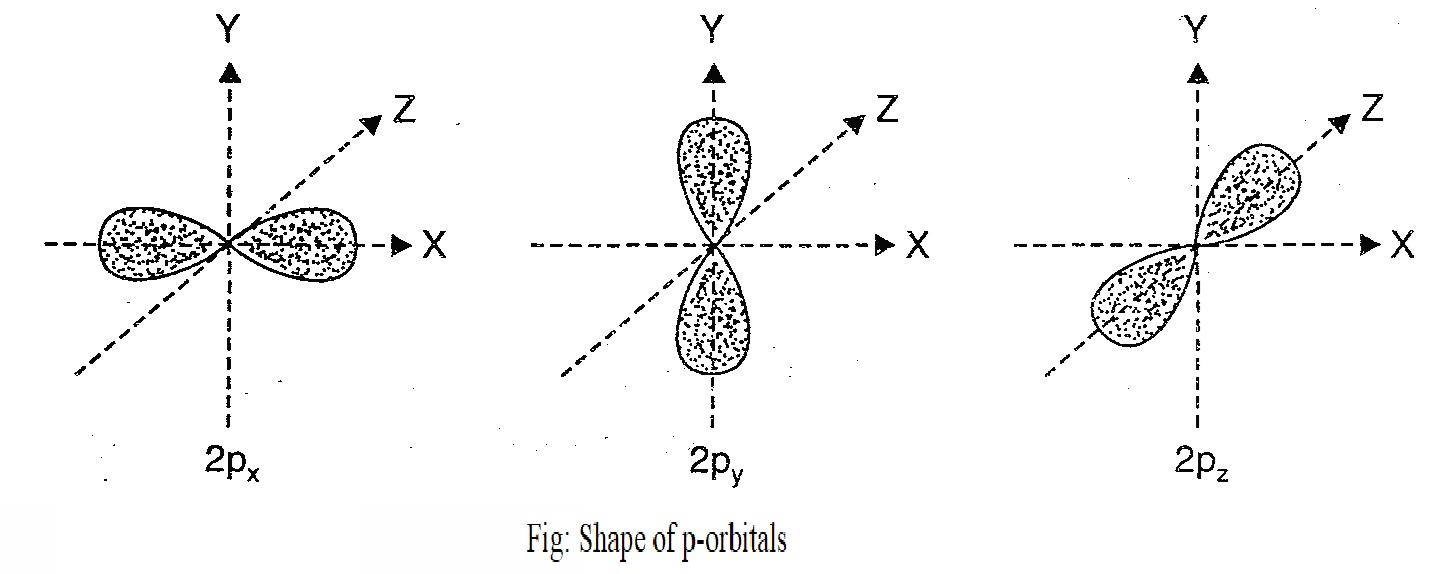 Shape of d-orbital:
Shape of d-orbital:
For d-subshell, l=2, there are five values of m viz. +2, +1, 0, -1, -2. It means d-orbital can have five orientations. These are represented by dxy, dxz, dyz, dz2 and dx2–y2 respectively. These five orbital are equal in energy (degenerate state), but differ in their orientation. The dxy, dxz, and dyz orbital have same shape but lie in different plane. The dz2 orbital is symmetrical about z-axis and has a dumb-bell shape with doughnut shape electron cloud in the centre. The dx2–y2 orbital is also clover leaf shaped and lies along x and y axis.

Fig: Shape of d-orbital.