Solubility Product :
When a sparingly soluble salt is dissolved in water then even in the saturated solution, the concentration of the salt is very low. So, whatever little of electrolyte goes into solution undergoes complete dissociation (due to low concentration). Therefore, in saturated solutions of such electrolytes, the solid electrolyte is in equilibrium with the ions as represented below:
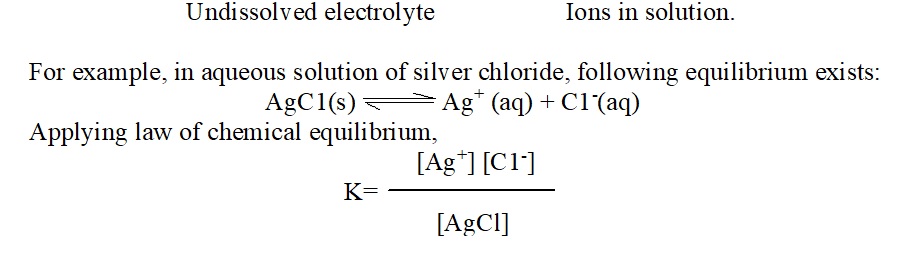
The concentration of the undissolved salt is constant (say k) at a particular temperature regardless of the amount of solid silver chloride present. Therefore, we have
K × [AgCl] = [Ag+] [C1–]
Or, Ksp = [Ag+] [C1–]
The constant Ksp is known as solubility product. It is equal to the product of ionic concentrations, termed as ionic product, for a saturated solution. The solubility product constant, Ksp, is the equilibrium constant for the solubility equilibrium of a sparingly soluble ionic compound. Therefore, “The solubility product of a sparingly soluble salt at a given temperature is the product of the concentrations of its ions in the saturated solution, with each concentration term raised to the power equal to the number of times the ion occurs in the equation representing the dissociation of the electrolyte”.