Half-life period (t½):
Half life period is the time during which the concentration of reactants is reduced to half of the initial concentration. It is denoted by t½.
1) Half-life period of a zero order reaction:
The integrated equation for zero order reaction is—
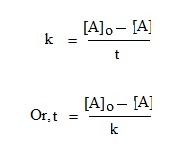 At t = 0, then [A] = ½ [A]o, Then above equation become
At t = 0, then [A] = ½ [A]o, Then above equation become
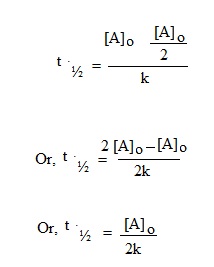 From the above expression it is clear that half life period of a zero order reaction is directly proportional to its initial concentration of reactants i.e.
From the above expression it is clear that half life period of a zero order reaction is directly proportional to its initial concentration of reactants i.e.
![]()
2) Half life period of a first order reaction:
The integrated equation for first order reaction is—
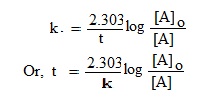
When t = t½, then [A] = ½ [A]o
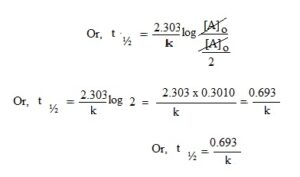
Therefore, the half life period of a first order reaction do not depend on the initial concentration of reactants.